Description
- The FlowFlex™ COVID-19 Antigen Home Test is a lateral flow immunochromatographic assay for the detection of extracted nucleocapsid protein antigens specific to SARS-CoV-2, not for any other viruses or pathogens.
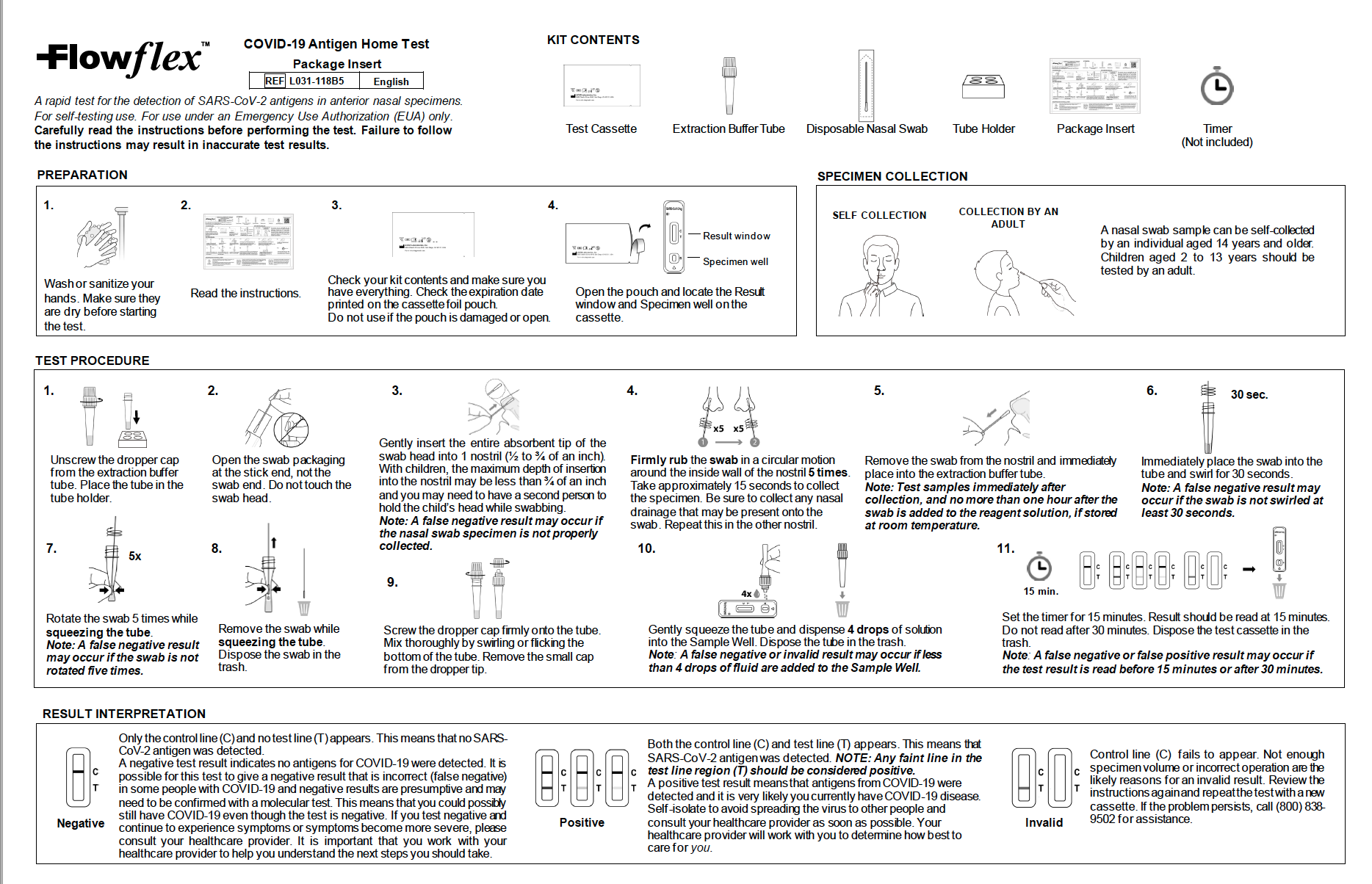
- For use with direct anterior nasal and nasopharyngeal swab specimens, non-invasive test procedures
- Detect SARS-CoV-2 nucleocapsid protein antigen
- Accuracy:
- Nasal Swab: 98.8% (95% CI: 97.6%-99.5%) 95% Confidence Intervals
- Saliva: 97.1% (95% CI: 94.6%-98.5%) 95% Confidence Intervals
- Variant-ready solution
- Rapid results within 15 minutes
- Expiration Date: If you receive a box that has an expiration date that has past, please do not be concerned. The state department has granted an extended expiration life by 12 months from the expiration date printed on the back of the box for these tests.
Kit Info
- 1 Test/Box
Contents Include
- 1 Nasal Swab
- 1 Test Cassettes
- 1 Extraction Buffer Tubes
- Packaging insert
FlowFlex™ COVID-19 Antigen Home Tests Provide Accurate Results
Because we need to prevent the spread of COVID-19, we must put in the effort to perform accurate testing on everyone who needs it. When you buy FlowFlex™ COVID-19 Antigen Home Test, you give people a reliable way to show they are in good health and can breathe easily in their environment, be it work or school.
The FlowFlex™ COVID-19 Antigen Home Tests are suitable for medical professionals and the public to use. When you use the test, you can see if you carry COVID-19 antigens. Testing for antigens is another way to determine if you contracted COVID-19, especially if you are asymptomatic. The test is easy to use and requires a quick nasal swab placed in a tube filled with a solution ready to react to the samples collected. The package comes with detailed instructions, so you will know exactly what to do.
FlowFlex™ is a rapid test. Get your results within 15 minutes and make those surrounding you aware of the outcomes, so they can make necessary preparations.
Disclaimer:
- This product has not been FDA cleared or approved; but has been authorized by FDA under EUA;
- This product has been authorized only for the detection of proteins from SARS-CoV-2,not for any other viruses or pathogens; and
- This product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. 360bbb-3(b)(1),unless the declaration is terminated or authorization is revoked sooner.